How did you find using Pulsar-18 T3 the first time?
Clinically Proven Thin Strut Stent Design



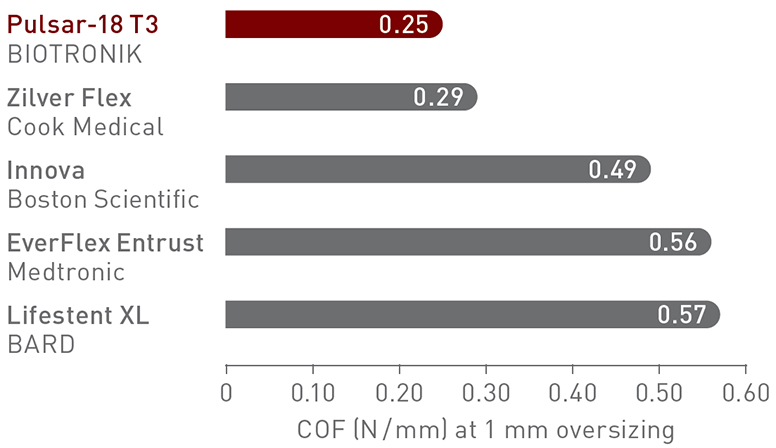
What is COF and why does it matter?
Chronic outward force (COF) is the force exerted on the vessel wall by a self-expanding stent to achieve its present diameter. Thinner struts, which have lower COF*, result in a lower risk of restenosis, reduced vessel injury and inflammation and faster endothelialization.2,7,8
140 μm thin struts - thinner than leading brands9
Thinner struts for lower COF10
Achieve less mean restenosis with
low-COF Pulsar-18 T311
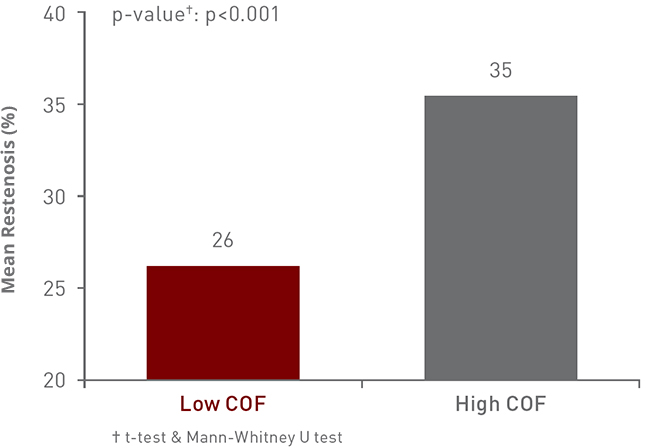
*as demonstrated in pre-clinical studies
Physician Testimonials
How does Pulsar-18 T3 meet your treatment objectives?
Easy to use, intuitive wheel-operated handle and one-handed deployment.
The first and only, 4F thin-strut stent system.


Contact us
Pulsar-18 T3 is indicated for use to improve luminal diameter in patients with symptomatic de novo, restenotic, or occlusive lesions located in the superficial femoral or proximal popliteal arteries, with reference vessel diameters from 3.0 to 6.0 mm and total lesion lengths up to 190 mm. As per IFU.
The product information presented here is intended exclusively for healthcare professionals. Prior to use, reference the Instructions for use online or inside the product packaging.
- BIOTRONIK data on file;
- Zhao HQ Late stent expansion and neointimal proliferation of oversized nitinol stents in peripheral arteries. Cardiovasc. Interv. Radiol. 2009; 32(4); 720-6.;
- Deloose K. 4EVER 24m: Long-term results of 4F Pulsar stents in femoropopliteal lesions. Presented at: LINC 2014; Jan 29; Leipzig, Germany;
- Lichtenberg et al. Effectiveness of the Pulsar-18 self-expanding stent with optional drug-coated balloon angioplasty in the treatment of femoropopliteal lesions - the BIOFLEX PEACE All-Comers Registry. Vasa (2019), 1-9. doi_10.1024/0301-1526/a000785; 2014; Leipzig, Germany;
- Bosiers M et al. 4-French – compatible endovascular material is safe & effective in the treatment of femoropopliteal occlusive disease: Results of the 4EVER Trial. ENDOVASCTHER 2013; 20: 746-75;
- Lichtenberg M. Superficial Femoral Artery TASC D registry: 12-month effectiveness analysis of the Pulsar-18 SE nitinol stent in patients with critical limb ischemia. J Cardiovasc Surg (Torino). 2013 ; 54(4):433-9;
- Koskinas C. Role of endothelial shear stress in stent restenosis and thrombosis: pathophysiologic mechanisms and implications for clinical translation. JACC 2012 10;59(15):1337-49;
- Koppara T. Thrombogenicity and early vascular healing response in metallic biodegradable polymer-based and fully bioabsorbable drug-eluting stents. Circ Cardiovasc Interv. 2015 8(6):e002427;
- BIOTRONIK data on file. 6.0 mm diameters;
- BIOTRONIK data on file. 6.0 mm diameters. Supera stent not possible to test due to its design and applied test method;
- Funovics M. Differences in clinical outcomes of low COF stent vs high COF stent proven in clinical practice. Presented at: CIRSE, Sep 8, 2019; Barcelona, Spain;
- Pulsar-18 T3 IFU.









